
Early pregnancy health
Good health and care in pregnancy are essential for you and your baby and are especially important in early pregnancy. This information brochure outlines the key issues in early pregnancy health and care, and what to do next now that you are pregnant.
When should I see a doctor?
Ideally, you should see a doctor before you become pregnant to discuss any tests you may need. You can also discuss any medical problems, medications, supplements, and general health in pregnancy. If this hasn’t happened because your pregnancy was unplanned or unexpected, then when you first find out you are pregnant is an excellent time to visit your GP. They can give you important information, answer questions, and discuss the essential routine blood tests in early pregnancy. They can also discuss options for care in pregnancy and refer you to whoever will be undertaking your pregnancy care.
How is the length of a pregnancy calculated?
Pregnancy is counted from the first day of your last normal menstrual period and lasts about 40 weeks from that time.
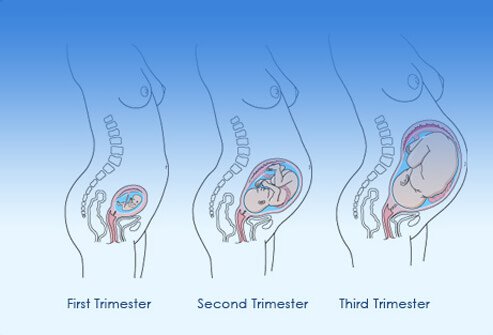
PREGNANCY is divided into 3 trimesters:
- First trimester: up to 12 weeks
- Second trimester: 13 to 27 weeks
- Third trimester: 28 to 40 weeks
Health in early pregnancy
Nutrition in pregnancy
Pregnancy places significant demands on your body. Good nutrition during this time is very important for your health and the health of your developing baby.
You only need a small increase in the amount of food or calories; it is the quality of your diet that really matters. Your need for protein, iron, iodine, and folate do increase in pregnancy.
However, what is most important is a healthy balanced diet that includes foods from all food groups. A healthy diet has a high intake of fresh fruit, vegetables, whole grains, proteins such as legumes, eggs, nuts and seeds, tofu, lean meats, fish, and calcium-containing foods such as dairy or calcium-fortified dairy alternatives. A vegetarian or vegan diet can meet the needs of your pregnancy as long as you take care to include enough protein, iron, vitamin B12, and calcium-containing foods. For further information on diet in pregnancy, talk to your GP or a dietician.
Folate
Folate (also known as folic acid) is one of the B-group vitamins and is essential for the healthy development of a foetus in early pregnancy. Folate reduces the incidence of neural tube defects (such as spina bifida) and some other serious disorders.
Folate is found naturally in foods such as green leafy vegetables, broccoli, brussel sprouts, oranges, bananas, strawberries, and legumes. However, most people do not eat enough to meet their pregnancy needs, and taking a supplement is always recommended.
Taking 500 micrograms of folate daily for one month before conception and during the first twelve weeks of pregnancy is advised. Folate is available in tablet form from any pharmacy. If you have certain conditions suchas diabetes or epilepsy, a higher dose of folic acid may be recommended for you.
Iodine
Iodine is an essential nutrient for humans. It is especially important for your baby’s developing brain and nervous system during pregnancy and in infancy. Therefore, it is recommended you take an iodine supplement of 150 micrograms a day during pregnancy and breastfeeding. Discuss this with your GP to make sure that you have no contraindications.
Iron
Iron is an essential nutrient needed to make red blood cells that carry oxygen around your body. Your iron requirements increase during pregnancy and a deficiency can lead to tiredness, dizziness, and lower resistance to infection. You can usually get enough iron from eating iron rich foods, which is recommended rather than taking a supplement. However, some people may find that they need iron supplementation, such as those who have a vegetarian or vegan diet, have a history of iron deficiency, or have a multiple pregnancy. Iron supplements can cause side effects, so discuss this with your GP if you think you may need one.
Vitamin D

The most important source of vitamin D is sunlight.
Vitamin D is a nutrient that helps maintain bone and muscle strength, and assists you with absorbing calcium. It also helps your baby to develop strong bones. The most important source of vitamin D is sunlight. In summer, average daily exposure to sunlight should supply adequate vitamin D without putting you at increased risk of skin cancer. In winter, the recommended exposure is 2-3 hours a week. Darker-skinned people need longer exposure to sunlight to get enough vitamin D. Taking a vitamin D supplement in pregnancy is not recommended unless you have low vitamin D levels, and your GP advises it.
Vitamin B12
Vitamin B12 is one of the B group vitamins and is found in meat and eggs. It is essential for the development of the brain and nervous system of your baby. Some people are at an increased risk of B12 deficiency, including anyone with a family history of B12 deficiency, with a condition affecting absorption, or if you have a strict vegetarian or vegan diet. In this case, you should consider supplementation with Vitamin B12 in pregnancy and during breastfeeding. B12 is available in tablet form from any pharmacy. If you are at risk of B12 deficiency, discuss this with your GP.
Calcium
Calcium is an essential mineral and is important during pregnancy and breastfeeding because it is needed for your baby’s bones to form. Calcium supplements are not considered necessary in pregnancy unless advised by your GP. Just make sure that you are getting enough calcium as part of a healthy diet. Dairy foods, such as milk, cheese, yoghurt, and calcium-fortified soymilk are excellent sources of calcium. You should aim for three serves a day.
Multivitamin supplements
While there is some evidence that taking a multivitamin in pregnancy may reduce the risk of a baby being small, the risk of pre-term birth, and of some abnormalities, a healthy balanced diet should be adequate for most people. If you still wish to take a multivitamin discuss this with your GP, midwife, or obstetrician.
Herbal preparations
There is not enough evidence on the safety or effectiveness of using herbal preparations during pregnancy. Ginger may help to reduce pregnancy-related nausea and is considered relatively safe. You should avoid taking any other herbal preparations in the first trimester. If you are considering taking any herbal preparations other than ginger, please talk to your GP.
Mercury In fish
Fish can be a valuable source of protein in pregnancy. However, some fish contain mercury which can cause developmental delays in children exposed during pregnancy. It’s best to limit the amount of fish you eat which may contain a high level of mercury during your pregnancy. Fish that contain higher levels of mercury include flake, ray, swordfish, barramundi, gemfish, orange roughy, ling, and southern blue fin tuna.
Water

Your need for water increases significantly during pregnancy. This is because of the changing needs of your body and your baby’s needs, so it is very important to make sure that you are drinking plenty of water every day.
Caffeine
While there is no clear evidence of harm from consuming caffeine in pregnancy, some studies have linked high caffeine consumption with miscarriage and low birth weight in babies. Low to moderate caffeine consumption (up to 3 cups of coffee or 6 cups of tea a day) is considered safe in pregnancy. Be aware that cola drinks contain caffeine too. Energy drinks have a high caffeine content and should be avoided.
Weight gain in pregnancy
Weight gain in pregnancy is very normal, and is due to changes in your body tissue, increase in the size of your breasts and uterus, increases in fluid and blood volume, and the weight of your developing baby. However, the amount of weight gain recommended will depend on your pre-pregnancy weight.
Weight gain varies between individuals. Average weight gain is usually between 11.5 and 16 kilos. Most pregnant people gain around 1 to 2 kilos in the first 3 months, and 1 to 2 kilos a month for the last 6 months. It is important not to try to lose weight in pregnancy as this can affect the health of the baby, however gaining too much weight can also cause health problems for you. If you are concerned about weight, discuss this with your GP or with a dietician.
If you are underweight, additional serves of the five food groups may contribute to healthy weight gain. If you are overweight or obese, limiting additional serves and avoiding energy-dense foods may limit excessive weight gain. Weight-loss diets are not recommended during pregnancy.
Food safety in pregnancy
Your immunity is lower in pregnancy and there is a slightly increased risk of developing a foodborne illness. These illnesses include listeria, salmonella, campylobacter, and toxoplasmosis and can be dangerous for your baby. It is important that you take steps to reduce the risk of becoming infected.
Avoiding foodborne illnesses during pregnancy
Eat mainly freshly prepared and cooked foods and:
- Avoid chilled seafood such as smoked salmon, mussels, oysters, prawns, or raw seafood such as sushi
- Avoid pre-prepared or pre-packaged food from buffets, salad bars, and sandwich bars
- Be cautious with deli meats such as pate, ham, salami, or pre-cooked and diced chicken
- Avoid undercooked meat or poultry
- Avoid unpasteurised milk or foods made from unpasteurised milk
- Avoid soft-serve ice-creams
- Avoid soft cheeses, such as brie, camembert, ricotta, and feta (these are safe if cooked and served hot). Hard cheeses such as cheddar are fine
- Avoid ready-to-eat foods, including leftover meats, which have been refrigerated for more than one day
- Avoid dips, and salad dressings in which vegetables may have been dipped
- Avoid pre-cut and pre-packaged vegetables or fruit, or make sure you cook them before eating
- Do not eat raw sprouts • Only eat cooked eggs and avoid products made with raw eggs, such as mayonnaise or aioli

Practice good food/hand hygiene
Follow good food handling practices such as regular handwashing, washing fruit and vegetables before eating or cooking, keeping surfaces and cutting boards and implements clean, and keeping raw meat separate from other foods.
Toxoplasmosis
This is a parasite typically found in birds and mammals (mainly cats). It is also found in raw pork and lamb. Infection during pregnancy can cause congenital disabilities in babies. Avoid close contact with cats, do not change litter trays, wear gloves during gardening, and avoid uncovered sandpits where you might find cat faeces.
Exercise in early pregnancy
Low to moderate intensity exercise in pregnancy can be very beneficial and has range of health benefits. However, it is advisable to avoid any physical activity that may risk a fall, any abdominal trauma, or excessive strain on your joints. These include high-impact sports and contact sports. Scuba diving is also not advised. If you have any physical problems or conditions that may interfere with exercising always discuss it with your GP or midwife first.
Medications/drugs
While some medications are quite safe to use in pregnancy, many medications and prescription drugs can be harmful to unborn babies. You need to discuss any prescription medications with your doctor if you are thinking of becoming pregnant, or as soon as you are pregnant.
Do not take any over-the-counter medications without checking with your doctor, pharmacist, or midwife. This includes common drugs such as aspirin or ibuprofen, as well as any complementary or herbal medicines. It is usually better to avoid taking any non-essential medication; however, some medications such as antibiotics are necessary and safe options can be prescribed.
It is important not to stop vital medications without consulting your doctor. For example, asthma medications are safe in pregnancy, and it is important that asthma is well controlled when you are pregnant.
Alcohol
Alcohol consumed during pregnancy passes directly into your unborn baby’s blood supply. It can have severe effects on an unborn baby and can result in a condition called foetal alcohol spectrum disorder (FASD), which has a range of serious physical, behavioural, and mental effects. The safe level of alcohol during pregnancy is unknown and even a small amount at the wrong time can cause problems. For this reason, you should avoid drinking any alcohol during pregnancy or when you are planning pregnancy.
Illicit drugs
Many illicit drugs can seriously affect your baby’s health if used during pregnancy. These include marijuana, MDMA, cocaine, amphetamines, LSD, and heroin. It is best to avoid all drugs during pregnancy. However, if you are using any drugs be sure to talk to your GP or midwife to help you and your baby stay as healthy and as safe as possible.
Smoking
It is now well known that smoking causes serious harm to you and your developing baby. This includes passive smoking, where someone is exposed to another person’s smoking. Smoking in pregnancy can cause complications and is linked with pre-term birth, a baby having a low birth weight, being small for age, some birth abnormalities, and even an increased risk of miscarriage, stillbirth, and SIDS.
The sooner that you stop smoking in pregnancy the better for you and your baby.
TALK TO YOUR GP ABOUT GETTING HELP TO STOP OR REDUCE SMOKING OR CALL THE QUITLINE ON 137 848.
Environmental toxins
Some common substances are known to be potentially toxic during pregnancy and are best avoided completely, these include dry cleaning products, oven cleaners, paint thinners or paint strippers, Insecticides, herbicides, and any products or materials which may contain lead or mercury. If you are uncertain about a substance, then it is best to avoid it..
Morning sickness
Morning sickness during pregnancy is common. Symptoms include loss of appetite, aversions to certain foods and smells, nausea, and vomiting. Despite its name, morning sickness can occur any time of the day or night. You can do things to lessen the effects of morning sickness. For example, avoiding an empty stomach. You can do this by eating very small frequent meals, eating a few dry crackers before getting out of bed in the morning, and keeping light snacks with you to eat during the day. Avoiding any rich, fatty, or spicy foods is also helpful.
Occasionally morning sickness can become severe, and in some cases, may require hospitalisation. If you experience persistent vomiting, see your GP. Medications that are considered safe to use in pregnancy are available to help reduce morning sickness.
Travel in pregnancy
Car travel
Car travel carries a known risk for everyone. Safe driving advice applies as usual. Seat belt use reduces the risk of serious outcomes for you and your baby in the event of an accident. Therefore, it is important that you always wear a seatbelt, and to make sure the straps are placed above and below the bump, not over the bump.
Air travel
Air travel before 36 weeks of pregnancy is generally considered safe. As air travel can increase the risk of blood clots, take the usual precautions of moving about when you can, staying well hydrated, and using support stockings to reduce this risk. Make sure the lap seat belt is worn under the bump. Most airlines restrict travel for people after 36 weeks of pregnancy.
Overseas travel
Immunity is lower in pregnancy. There is a slightly increased risk of developing a food-borne illness that can be harmful to your baby, so caution should be used when traveling to countries with a high rate of food-borne illnesses. Anti-malaria medication is not safe during pregnancy, so travel to malaria-endemic countries is not advised if you are pregnant. If travel to these areas is unavoidable, then other precautions should be taken to avoid bites from malaria-carrying mosquitoes. Discuss this with your doctor or a specialist travel clinic.
Travel vaccines
If you are traveling when pregnant make sure that you have travel insurance. Any travel insurance policy you purchase needs to cover you for pregnancy, and for pregnancy or birth complications.
Travel insurance
If you are traveling when pregnant make sure that you have travel insurance. Any travel insurance policy you purchase needs to cover you for pregnancy, and for pregnancy or birth complications.
Sex In Pregnancy
Sexual activity in pregnancy is considered quite safe if there are no complications with the pregnancy. Libido in pregnancy is quite individual; some people’s interest in sex decreases, but it may be unchanged or even increase for others. It is common for interest in sex to decline as the pregnancy progresses.
MENTAL HEALTH

Pregnancy and the experience of pregnancy is individual for everyone. Some people find that their mental health remains unchanged or that they may even feel calmer than usual during pregnancy. However, some people experience increased anxiety and/or depression during pregnancy or in the post-natal period. If you have had mental health concerns before pregnancy then your risk of developing a problem during pregnancy is slightly increased.
If you have concerns about this or feel that you are experiencing some changes to your mood or to your level of anxiety, it is very important to discuss this early with your GP or midwife. There is good support available.
- ACT Health runs a support line for anyone who needs support in pregnancy, Early Pregnancy Support: Call 02 5124 1775.
- Perinatal and infant Mental Health. This is a free service run by Canberra Health Services for people who are pregnant or have a baby under 12 months and have a severe mental health problem. It is best accessed through your GP or midwife. You can also self-refer. Call 02 5124 3133.
- The Perinatal Wellbeing Centre is a non-government organisation supported by ACT Health that provides individual and group mental health support to people in pregnancy and after birth until your youngest child is 2. The service is free, and you do not need a referral. Call 02 62881936
- If you are a client of Mental Health ACT or receiving opioid replacement there is a special service called the IMPACT Program that can assist you with managing your care in pregnancy. You can access the service by phone on 1800 211 274 from 9:00am to 4.30 pm weekdays.
- There are also some private psychologists who specialise in perinatal mental health.
If you have a serious diagnosed mental health issue such as bipolar disorder, borderline personality disorder, schizophrenia, or major depressive disorder you need appropriate care and support during your pregnancy. Talk to your GP, midwife, or your mental health professional about your pregnancy and the support services available for you.
Higher risk pregnancies
People may sometimes need specialised care in pregnancy. If you have a serious medical condition or have had a previous pregnancy affected by blood clots, pre-eclampsia(high blood pressure), pre-term birth, or a previous still birth you are considered at higher risk in your next pregnancy and are advised to see your GP early so that appropriate care can be arranged.
Pregnancy & birth care
Pregnancy care and birthing options
Care during pregnancy is called antenatal care. Regular antenatal care is important for your health and the health and wellbeing of your baby.
Your GP, midwives, an obstetrician, or a combination of these can provide antenatal care. The recommended number of visits for antenatal care during pregnancy is eight to ten.
Your first antenatal visit should include:
- a family history,
- a medical check, including routine blood tests
- a discussion about your pregnancy and birth care
- discussions about tests in pregnancy
It is a good idea to start thinking about the kind of care you want to receive during pregnancy and birth as soon as possible and to discuss this with your GP. There are several choices available in the ACT.
Models of care include midwife-led continuity of care programs (available through the Centenary Hospital for Women and Children or North Canberra Hospital) or care with a private obstetrician. In addition, specialist care is available for higher risk pregnancies.
There is also a publicly funded homebirth service for those who meet the criteria, and private homebirth midwives.
If you have private health insurance and are considering seeing a private obstetrician, make sure your plan covers pregnancy and delivery.
For more information on pregnancy care and birthing options in the ACT see the websites below.
Early pregnancy information and birth education classes

Most hospitals offer free antenatal and birth education classes to provide you with current information and education about pregnancy, birth, and breastfeeding. There are also private classes available. For more information see: Having a Baby in Canberra and ACT Health: Pregnancy and Birth (websites on next page).
FOR MORE INFORMATION ON BIRTH OPTIONS, CLASSES & EDUCATION IN CANBERRA VISIT 'HAVING A BABY IN CANBERRA'.
WHAT SHOULD I DO NEXT?
The next step is to make an appointment to see your GP, or find a GP if you don’t have one.
RECOMMENDED WEBSITES
For reliable evidence-based health information on pregnancy, birth and beyond see:
- ACT Health: Pregnancy & Birth
- Having a Baby in Canberra
- Health Direct Australia: Pregnancy
- The Royal Hospital for Women
FOR OTHER HEALTH SERVICES AND INFORMATION This email address is being protected from spambots. You need JavaScript enabled to view it. OR CALL 02 6247 3077
References: Department of Health (2020) Clinical Practice Guidelines: Pregnancy Care. Canberra: Australian Government Department of Health. Department of Health (2021) Who can get vaccinated for COVI-19 health.gov.au/initiatives-and-programs/covid-19-vaccines/getting-vaccinated-for-covid-19/who-can-get-vaccinated-for-covid-19. Foodsafety.gov: foodsafety.gov/risk/pregnant/index.html RANZCOG ranzcog.edu.au/Womens-Health. The Royal Women’s Hospital: thewomens.org.au
- Created on .
- Last updated on .